临床需求为本
技术创新为优
同筑患者福祉

关于我们
药欣生物成立于2016年,以临床需求为导向,围绕“成药性+创新药”的核心策略,多项产品进入临床阶段。核心团队由美国知名药企科学家、专家顾问组成,具有丰富的全球商业价值判断与渠道资源整合能力,致力于风险易控、前景可期的差异化“创新药+改良型创新药”研发与产业化。 公司秉持“临床需求为本,技术创新为优”的理念,凭借在难溶性药物口服制剂开发领域的技术优势,依托集科研转化、知识产权、注册法规为一体的“中美跨境创新药开发平台”,开发全球新药,推动国际前沿技术理念与科研成果高效转化,同筑患者福祉,共见健康未来。



我们的业务
药欣生物专注于以解决临床需求,提高患者预后为向导的新药开发。我们围绕临床需求解决关键问题,形成了先进的制剂技术和平台。基于这些具有显著竞争优势的关键平台技术,我们在新药和制剂改良开发方面形成了独特的产品布局,面向全球市场,开发符合FDA标准的创新药和改良型创新药,同时合作或引进具有潜力的CMC产品开发项目。目前,公司重点致力于肿瘤、神经系统、免疫系统疾病及过敏等领域。
创新药开发
公司依托在复杂制剂开发、成药性评估及CMC研究领域的系统能力,与早期新药发现企业(包括AI制药企业),筛选具备差异化优势、并能够充分发挥公司技术平台与资源协同效应的创新分子,共同推进面向中美双报的一类创新药研发。
改良型新药开发
(1)新剂型开发:
依托先进的制剂技术平台和专利技术,开展具有特色和技术壁垒的高难度改良型新药研发,提升目标药物的生物利用度、安全性及患者用药依从性。
(2)新适应症开发:
依托国际化的科学顾问和BD团队,发挥科学转化和项目评估体系优势,围绕未满足临床需求推进新适应症新药开发。
技术平台
药欣生物技术优势


独特的科学转化项目评估体系和系统化开发策略,结合国际领先的药物制剂开发平台技术,公司聚焦临床价值导向,持续推进创新药和改良型新药研发,构建差异化明显、具备全球竞争力的产品体系。
核心技术平台
新一代无定形固体分散技术(SprāSol®)
SprāSol®是药欣生物应用喷雾干燥技术自主开发的新一代无定形固体分散技术,已获 30 余项专利保护。该平台通过优化多组分配方与特色工艺设计,结合特殊的喷雾干燥工艺,首创在动力学和热力学层面构建有利微环境,形成物理稳定的无定形固体分散体系,可显著提升高熔点、高 LogP 值等难溶性化合物的体内吸收。相较传统技术,SprāSol® 对难溶性小分子及PROTAC等偶联小分子化合物具有更广泛的适用性和更优的吸收改善效果。
SprāSol®技术平台已建立从毫克级到公斤级以上的系统开发与放大能力,可支持中美双报临床样品制备及商业化生产技术转化,已有多个项目进入临床阶段。
处方前研究平台
药欣生物具有丰富的新药开发经验,能够针对处方前研究中的关键技术挑战提供系统解决方案,并将国际领先的制剂技术应用于新药开发。公司建有完善的固态分析检测平台,配备TGA、DSC、Xrpd、电子显微镜、激光粒度仪等专业设备,为药物开发提供坚实的研究与技术支持。
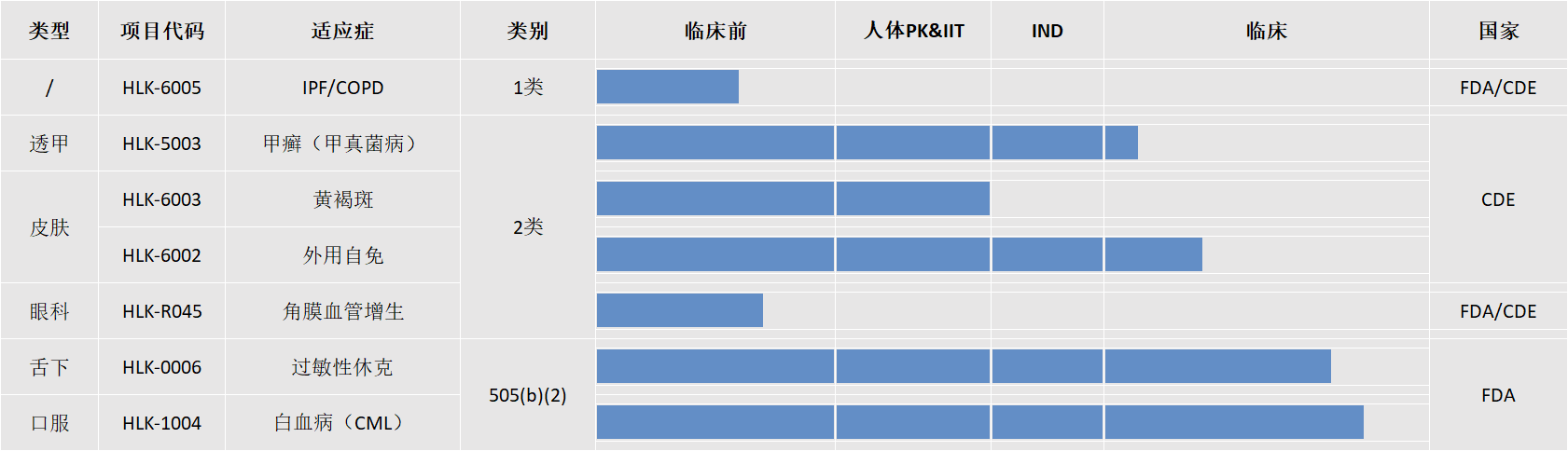
产品管线
研发产品管线
依托先进的制剂技术平台,公司聚焦临床未满足需求,持续推进创新药和特色制剂研发,已形成覆盖多个治疗领域的产品管线布局。
商业模式
创新,让灵感变成新药
面向全球市场,通过合作开发、项目引进等方式,开发具有差异化的一类创新药项目
开发符合中美监管标准的505(b)(2)新药和改良型新药
技术/产品授权转让或者联合开发

* = 必填
联系我们
我们欢迎各方创新思维与合作机会,共同应对药物开发挑战。药欣生物期待与合作伙伴携手前行、共创未来。如您有合作意向,欢迎垂询。
HLK Pharmacin, LLC
470 James Street
Suite 007
New Haven, CT 06513 USA
203-267-3309












